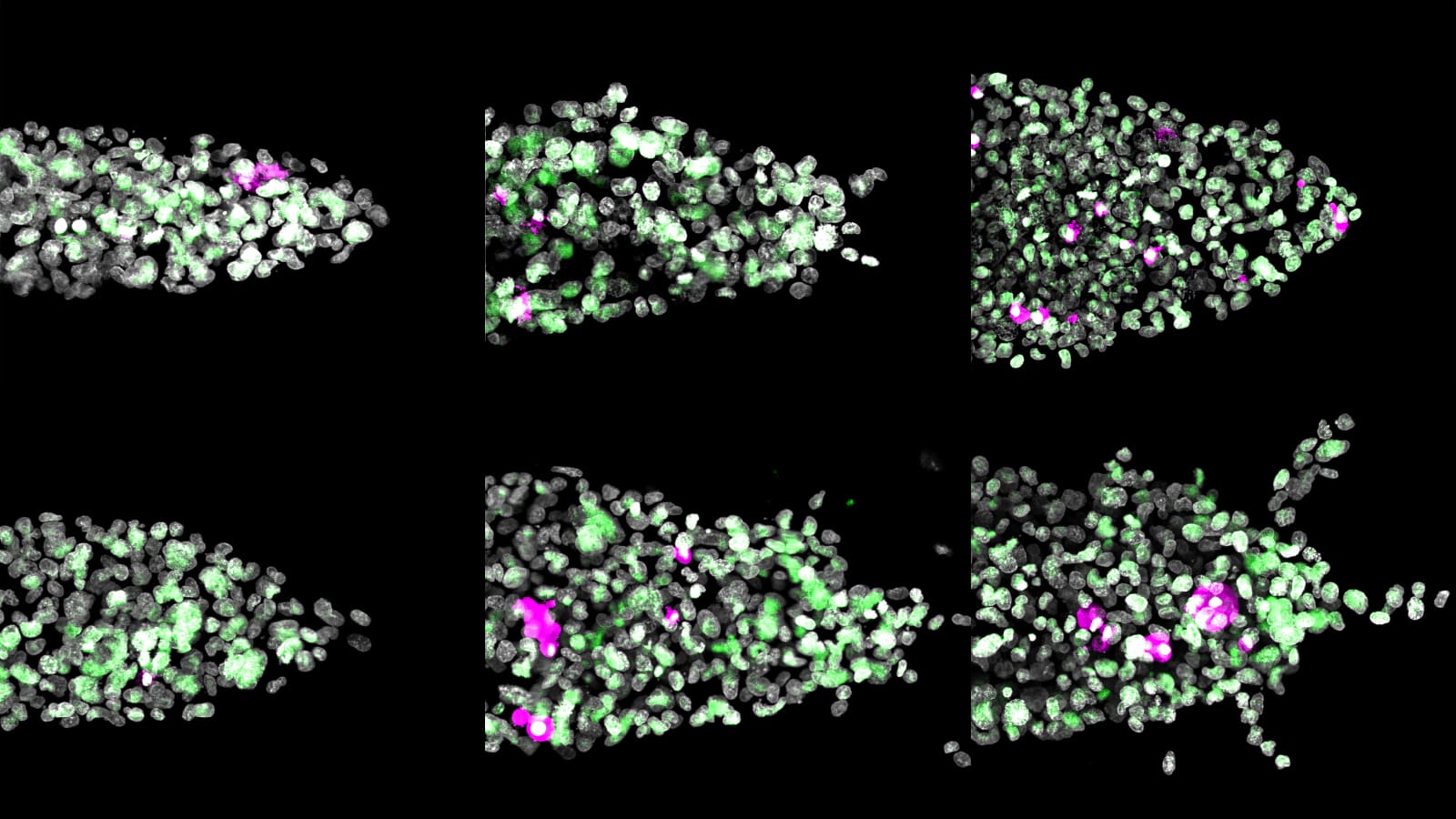
Biologists have long been fascinated by the first moments when cells divide to become complex
tissues and organisms. Now engineers — with an eye toward treating cancer and regenerating tissue
— are increasingly joining the hunt for the quantitative principles and underlying mathematics
that determine how these processes succeed or fail.
Stanislav”>http://www.princeton.edu/cbe/people/faculty/shvartsman/”>Stanislav
Shvartsman Ph.D. ’99, a professor of chemical”>http://www.princeton.edu/cbe/”>chemical
and biological engineering who also holds an appointment in Princeton’s Lewis”>http://www.princeton.edu/genomics/”>Lewis
Sigler Institute for Integrative Genomics, is developing statistical approaches to understanding
the way chemicals spread signals across an embryo. Shvartsman and colleagues published a breakthrough in
this work Oct. 17 in the journal http://dev.biologists.org/” target=”_blank”>Development.
In the lab of Celeste”>http://www.princeton.edu/~cmngroup/”>Celeste Nelson,
assistant professor of chemical and biological engineering, Cecillia Lui ’11 recently turned her
senior thesis into a peer-reviewed article on the mechanics of stem-cell differentiation in breast tissue,
which could have implications for understanding breast cancer. Another former undergraduate, Jay Kwak ’09,
is co-author with Nelson of a new study revealing that normal lung-tissue development is governed by a
single mathematical equation.
In the same department, Assistant Professor Clifford”>http://www.princeton.edu/cbe/people/faculty/brangwynne/”>Clifford
Brangwynne focuses on a cell component called the nucleolus, a loose confederation of proteins and
RNA that produces the hardware that builds cells from the inside out. A long-term goal is to tune the
properties of this tiny bioreactor to adjust cell growth, which could be useful in battling the runaway
expansion that characterizes cancer.