Although known since the 1830s as a round, dark spot in a cell’s nucleus, only recently has the nucleolus gotten its full due. Scientists have learned that besides building a cell’s protein factories, this specialized subunit, or organelle, serves more broadly as a control center for cellular growth and health.
In the last several years amidst a flurry of research, Princeton’s Clifford Brangwynne and colleagues discovered that the nucleolus behaves like a liquid with the consistency of honey. Yet somehow, this biological droplet maintains a complex, compartmentalized internal structure. Brangwynne, an assistant professor of chemical and biological engineering, and other researchers have puzzled over how such a liquid-like object could develop stably compartmentalized layers, rather than just fusing into a homogenous glob.
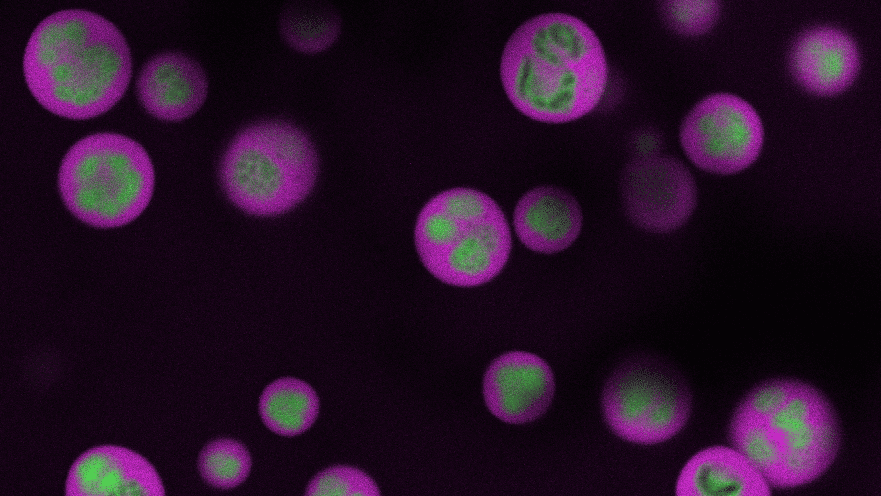
A new study by Brangwynne, his students and their collaborators presents a solution to the paradox of nucleolar assembly and internal organization. Their paper, published online May 19 in Cell, shows that the constituent proteins and RNA of nucleoli spontaneously assemble themselves into three distinct, liquid layers, thanks to their differing properties such as surface tension and viscosity. Rather like how oil and water can coexist yet remain separate, the nucleolus develops liquid subcompartments, which enable its critical functions. These insights into the nucleolus’ form and function could ultimately point toward new ways to treat disease.
“To get the characteristic layered, ‘core-shell’ architecture of the nucleolus, all you need to do is mix the right molecules together at sufficient concentrations,” said study co-lead author Marina Feric, a Ph.D. student in chemical and biological engineering.
“We’ve provided a biophysical mechanism for the structure of the nucleolus that automatically emerges from the collective behavior of immiscible liquids,” said the study’s other co-lead author, Nilesh Vaidya, a Helen Hay Whitney Postdoctoral Fellow in Brangwynne’s lab.
Brangwynne and colleagues had previously shown that a number of other organelles besides the nucleolus are also phase-separated liquid droplets of RNA and protein. The present findings point to how these organelles, all of which lack the obvious compartmentalization offered by membranes, can nevertheless exhibit intricate architectures tailored for their cellular duties.
“The basic properties of fluids dictate which components of the nucleolus are on the inside and the outside,” said Brangwynne. “Since the fundamental principles underlying this effect are observed even in non-living states of matter, we think this physical picture applies to many organelles inside cells.”
Other Princeton co-authors on the study are graduate student Lian Zhu and junior Tiffany Richardson, both in Brangwynne’s Soft Living Matter Group. Other authors include: Tyler Harmon and Rohit Pappu of Washington University in St. Louis; Diana Mitrea and Richard Kriwacki of St. Jude’s Research Hospital, Memphis. Funding for the research was provided in part by the National Science Foundation, the National Institutes of Health, the National Cancer Institute and the Helen Hay Whitney Foundation.
The researchers investigated nucleoli by conducting experiments with purified nucleolar proteins as well as living frog egg cells, roundworms and cultured cells, plus some computer modeling.
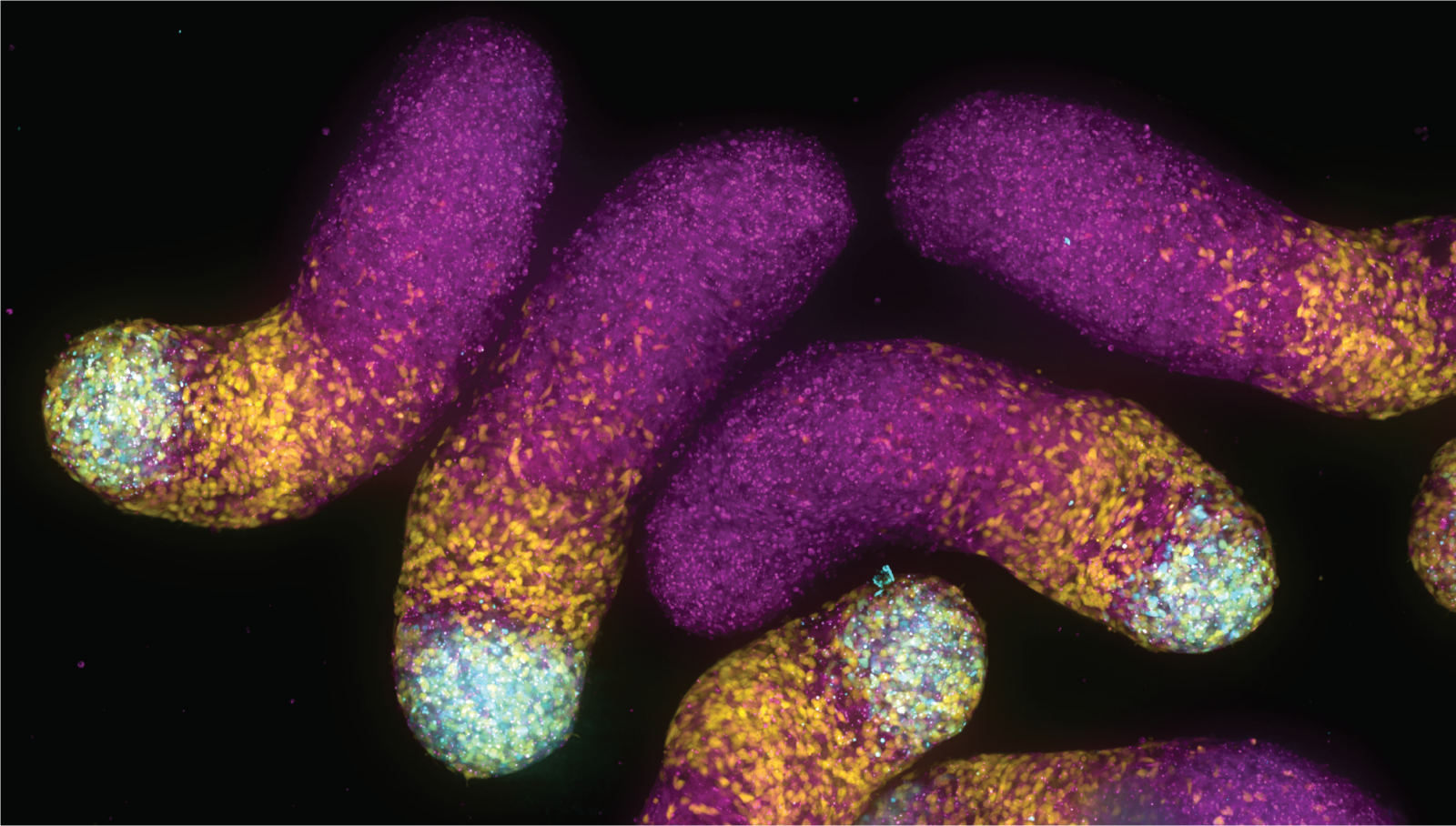
The work on the frog eggs – spearheaded by Feric – took advantage of the fact that the eggs possess multiple, large nucleoli, easing the organelle’s observation and manipulation. In these eggs, the nucleoli normally do not come into contact because of an elastic actin network. Feric incubated them in a pharmacological drug to break down the actin, allowing nucleoli to contact one another, and observed their contents fuse, much like how two droplets of water coalesce into a larger drop. By analyzing these fusion events, the researchers teased out the differing biophysical properties for the respective nucleolar layers.
Meanwhile, the protein-based lab work, led by Vaidya, found that droplets of the two main nucleolar proteins, dubbed FIB1 and NPM1, would not blend into each other as a homogenous fluid. Instead, because FIB1’s surface tension was higher than NPM1, the former became engulfed by the latter, precisely mimicking the nucleolar structure seen in living cells. As a demonstration of this phenomenon, the researchers also created similar multi-phase drops -liquids embedded within other liquids – using vegetable oil and silicone oil in water.
Altogether, the data handily explain how the individual subcompartments of the nucleolus nestle inside of each other, somewhat like a Russian matryoshka or “nesting” doll.
“This study is very important and exciting,” said Steve Michnick, a biophysical chemist at the University of Montreal who was not involved in the research. “It establishes both that the suborganization of the nucleolus results from one phase-separated body enveloping another and explains the physical complementarity between each body and the surroundings that results in this arrangement.”
For the nucleolus, this layered form follows function. Newly made RNA molecules proceed from the organelle’s core into the middle, then outer components, receiving modifications as they do so, as if on an assembly line. Like a factory, nucleoli churn out these RNA bits, which after leaving the nucleolus ultimately enter into the cell’s cytoplasm and link up to form structures called ribosomes. Factories in their own right, these ribosomes manufacture a cell’s many thousands of proteins.
Beyond making ribosomes, the nucleolus has lately emerged as a hub for coordinating cellular growth, helping to regulate cell division and even setting the timing of a cell’s self-destruction in reaction to stress or damage. Given this centrality, the nucleolus is also increasingly being recognized for roles in disease. For instance, during certain illnesses, nucleoli may lose some of their normal liquidness. If the faulty nucleoli become more fibrous, throwing off kilter the smooth flow of RNA through their chambers, this could contribute to heart and neurological diseases.
Another major disease nucleoli figure prominently in is cancer. In malignant cells, hijacked nucleoli overproduce proteins to fuel out-of-control, rapid divisions. Oncologists routinely grade cancer cells’ aggressiveness in part on the degree of their misshapen and bloated nucleoli.
Further insight into the normal layout of nucleoli, as the new Cell study provides, will thus help in assessing the organelle as a promising therapeutic target for drugs in near future. Brangwynne also hopes that understanding nucleolar core-shell structure will inform his lab’s ongoing studies into other overlooked organelles without membranes, like stress granules, p-bodies and Cajal bodies. Although these structures barely turn up in biology text books, recent work has suggested they too play key roles in both cell physiology and many diseases, including aggregation diseases such as Alzheimer’s and amyotrophic lateral sclerosis (ALS).
“This research is very exciting for us,” said Brangwynne. “It’s amazing how complex structures like the nucleolus can self-organize, based on relatively simple physical principles. We’re hoping that there is also simplicity underlying how such organization can go awry in disease.”