This article is from the Winter 2018 EQuad News.
“You try to answer fundamental questions,” said Brangwynne, an associate professor of chemical and biological engineering. “You hope they take you in interesting directions.”
Researchers in Brangwynne’s lab have moved into very interesting areas. In the past few years, they have explained how tiny structures called organelles grow and function within cells despite the lack of a surrounding membrane. The discovery that many organelles form through phase changes, like dewdrops appearing on grass, has led to a better understanding of cellular function. In one example, the research led the team to a way of understanding the growth of dangerous, solid-like protein aggregates inside the cells. These insights are shedding light on the proteins’ role in devastating degenerative neural diseases such as Alzheimer’s and ALS.
“Once you see how those pathological structures may be seeded, immediately you start thinking about ways to inhibit those processes,” he said. Brangwynne noted that a great deal of work needs to be done, but if all continues to go well, “potential pathways to therapies are going to emerge in the next few years.”
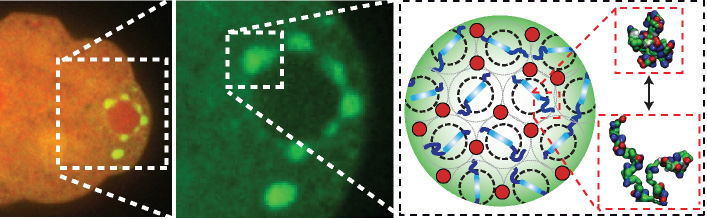
Last year, Brangwynne and his colleagues were able to engineer biological structures within cells. The technique, which also developed from basic research on organelles, opened up a possible path to enhancing certain cellular functions.
“From a basic ability to engineer structures within cells, we are starting to think about applications,” Brangwynne said. One possibility is to use engineered cells in industry for more efficient and faster protein purification, the laborious task of separating pure proteins from other cellular material.
Brangwynne said another possible track is enhanced production of valuable compounds within cells. The lab has already begun to collaborate with Jose Avalos, an assistant professor of chemical and biological engineering and the Andlinger Center for Energy and the Environment, on yeast strains that produce important natural products, such as forms of alcohol that could be used as fuel.
“I have been interested in biology for a long time, but I have been tackling biology through the lens of soft matter and materials physics,” Brangwynne said. “Some of the work we have been able to do is enabled by having a different perspective.
As an engineer, I see that the world has all these problems. So when we make basic discoveries about how cells work, I immediately start to think, ‘Why don’t we use this to solve some of them?’”