When Hippocrates first described cancer around 400 B.C., he referred to the disease’s telltale tumors as “karkinos” – the Greek word for crab. The “Father of Western Medicine” likely noted that cancer’s creeping projections mirrored certain crustaceans, and the tumors’ characteristic hardness resembled a crab’s armored shell.
Later, scientists added another attribute: Tumors are hypoxic. That is, they grow so large and dense that they exclude blood vessels, causing a lack of oxygen in their cores. But what role these characteristics play in the development of cancer has remained a mystery.
Moving one step closer to an answer, scientists from Princeton University and the Mayo Clinic Cancer Center have found that, in breast cancer, tumor hardness and hypoxia trigger a biological switch that causes certain cells to embark on a cancer-promoting program. Reported Aug. 8 in an article in the journal Cancer Research, this biological switch is critical to a tumors’ ability to invade other tissue, a process called metastasis – and could offer a promising treatment target.
“Our study suggests that to combat cancer, we should be developing treatments that target the stiff, hypoxic regions of tumors,” said lead author Celeste Nelson, a professor of chemical and biological engineering. “We were surprised to see just how important these two properties in the tumor microenvironment – stiffness and hypoxia – were for regulating cancer stem cells.”
[22].jpg)
The specific cells triggered by stiffness and hypoxia are called cancer stem cells. These cells represent only a small proportion of the total cells in a tumor, but researchers believe they play a key role in spreading the disease. As normal stem cells help form an embryo, or aid in repairing muscles, cancer stem cells specialize in generating new malignant cells. In addition to spreading cancer, just 10 to 100 leftover cancer stem cells are needed to regenerate a tumor after it has been removed.
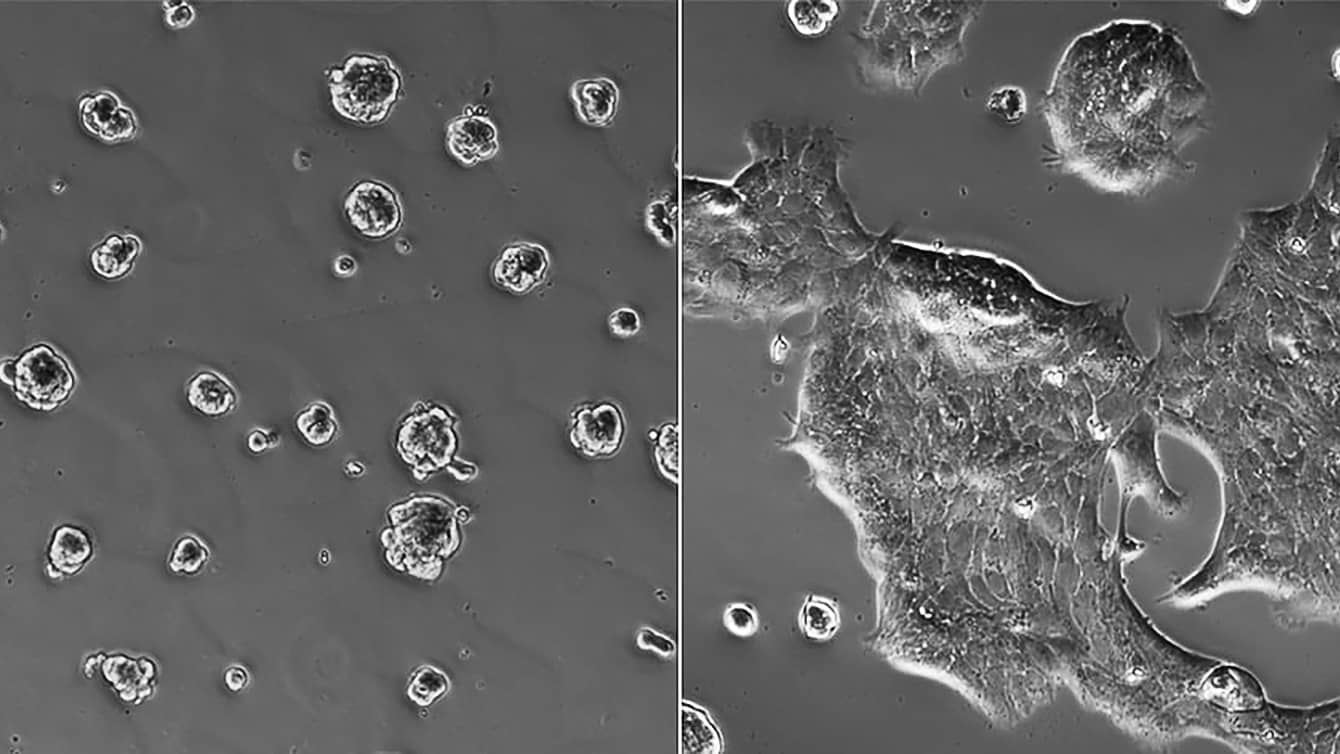
Using cultures of human breast-cancer cells and mouse mammary-cancer cells, Nelson and colleagues from Princeton and the Mayo Clinic in Jacksonville, Florida, discovered an association between a protein called integrin-linked kinase and the creation of cancer stem cells. Normally, integrin-linked kinase assists cells with a variety of important cellular tasks. But in dense, oxygen-poor tumors, the protein’s function goes awry.
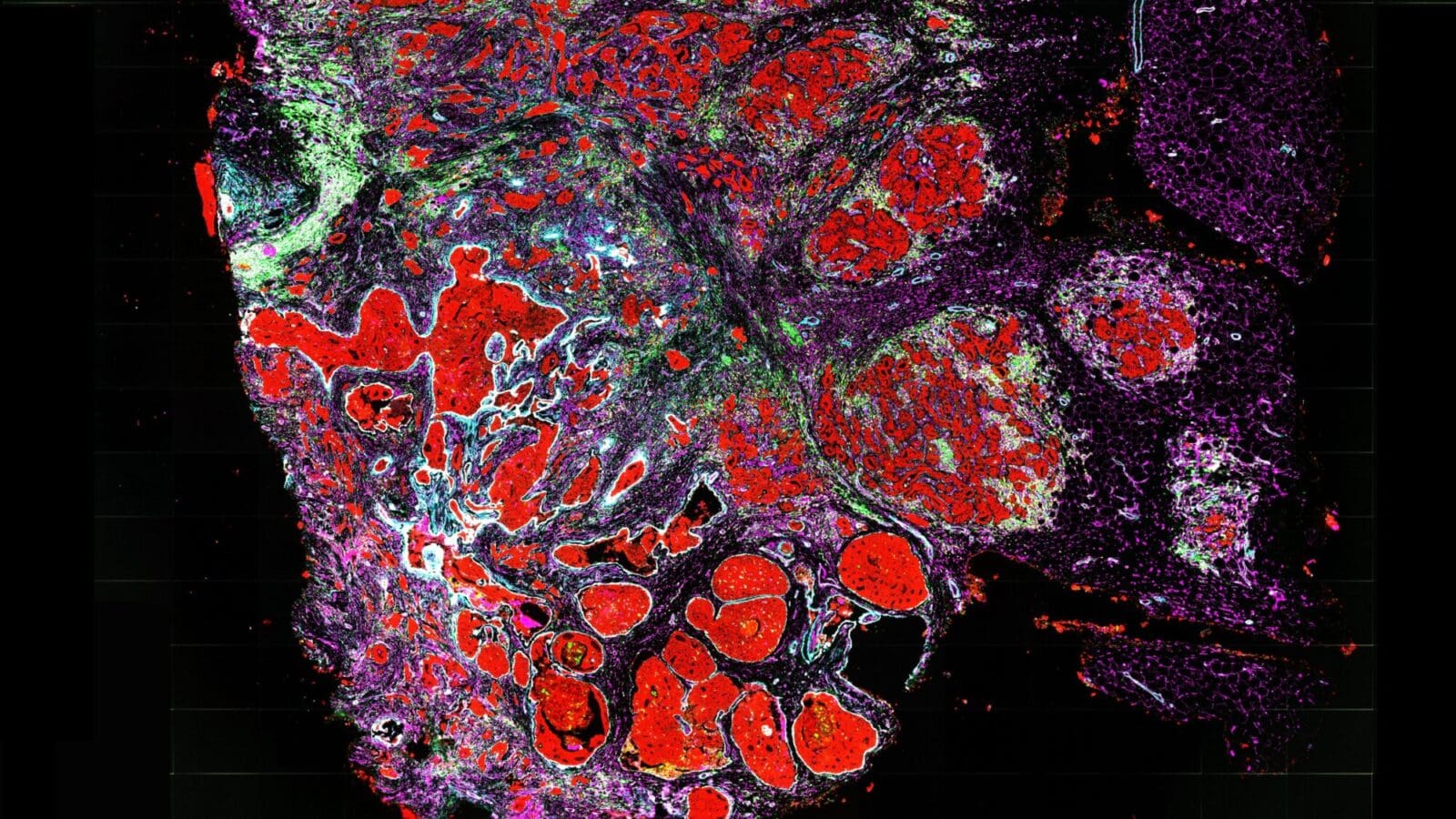
In the lab, the researchers created a range of human and mouse breast-cancer cultures reflecting different tissue conditions. They showed that stiff hypoxic cultures did indeed promote cancer stem cells. But when they eliminated the integrin-linked kinase from those samples, they found that the cancer stem cells stopped forming. Conversely, when they forced abnormal levels of integrin-linked kinase in samples containing softer or less hypoxic tissue, cancer stem cells formed. They also confirmed a significant association between tumor stiffness, integrin-linked kinase and cancer stem cell presence in samples from human breast-cancer patients.
“We could see tumor cells expressing cancer stem-cell markers and integrin-linked kinase located at regions with high collagen, which is used to estimate stiffness in a tumor,” says Mei-Fong Pang, a postdoctoral fellow in Nelson’s Tissue Morphodynamics Laboratory.
The findings suggest that stiffness and hypoxia cause integrin-linked kinase to behave abnormally, which in turn triggers cancer stem-cell formation.
There are likely other features in tumors that cause cancer stem cells to form, but the findings indicate that stiff, hypoxic conditions – and their effects on integrin-linked kinase – are two of the most prominent ones. This means the findings could be applicable for better understanding some types of cancer and for developing treatments for those characterized by solid tumors – including for more than just breast cancer.
“These findings may lead to the identification of a new therapeutic target to halt cancer progression and metastasis,” said Ren Xu, an associate professor at the University of Kentucky’s Markey Cancer Center who is familiar with the study but had no role in it.
“Given the crucial function of integrin-linked kinase in hypoxia and stiff-induced cancer progression, it is now critical to define the molecular mechanisms by which integrin-linked kinase expression is regulated under these conditions,” Xu said.
Nelson and her colleagues plan to investigate the specific molecular pathways that promote the formation of cancer stem cells in the presence of rigidity, hypoxia and integrin-linked kinase. Building on that knowledge, treatments could eventually be created that specifically kill cancer stem cells. Finding a way to change conditions in the tumor itself could provide another solution.
“If we can make the tumor softer or reduce hypoxia,” Nelson said, “we could potentially have a way to treat breast cancer and maybe other cancers as well.”
The paper, “Tissue stiffness and hypoxia modulate the integrin-linked kinase ILK to control breast cancer stem-like cells,” was published Aug. 8 in Cancer Research.
This work was supported by grants from the National Institutes of Health (grant nos. GM083997, HL110335, HL118532, HL120142, CA187692); the National Science Foundation (grant no. CMMI-1435853); the NSF Graduate Research Fellowship program; the David and Lucile Packard Foundation; the Alfred P. Sloan Foundation; the Camille and Henry Dreyfus Foundation; and the Burroughs Wellcome Fund; postdoctoral fellowships from the Swedish Society for Medical Research and the New Jersey Commission on Cancer Research.