Don’t throw away those bouncing batteries.
Researchers at Princeton University have found that common test of bouncing a household battery is not actually an effective way to check a battery’s charge.
“The bounce does not tell you whether the battery is dead or not, it just tells you whether the battery is fresh,” said Daniel Steingart, an assistant professor of mechanical and aerospace engineering and of the Andlinger Center for Energy and the Environment.
The battery bounce test, popularized in online videos, shows that fully charged batteries bounce very little when dropped, while those that have been used for a while bounce higher. The height of the bounce increases as the batteries discharge, and that has led to the common conclusion that internal changes related to the reduction in charge are the cause of the higher bounce.
“A year ago a buddy of mine who knows I work on this sent me this video and said did you know this happens?” Steingart said. “I didn’t. But I had a bunch of batteries on my desk and I was able to verify it.”
Steingart was intrigued by how the bouncing changed as batteries discharged – it was not a linear increase. Instead, the height rapidly increased and then leveled off. His research team has been working for some time on internal changes related to battery discharge, and he wondered whether the changing bounces reflected an important change in the batteries.
They devised a quick experiment in which they dropped a common battery through a plexiglass tube and used a computer microphone to record it striking a benchtop. The researchers then were able to use the time between bounces to determine the height of the bounce.

“What I really loved about this experiment is that the result holds a lot of scientific importance but it is also the kind of thing I can show to someone without a scientific background and they can still get something out of it.,” said Shoham Bhadra, a graduate student in electrical engineering and the Andlinger Center who is the lead author of the research paper reporting the findings.
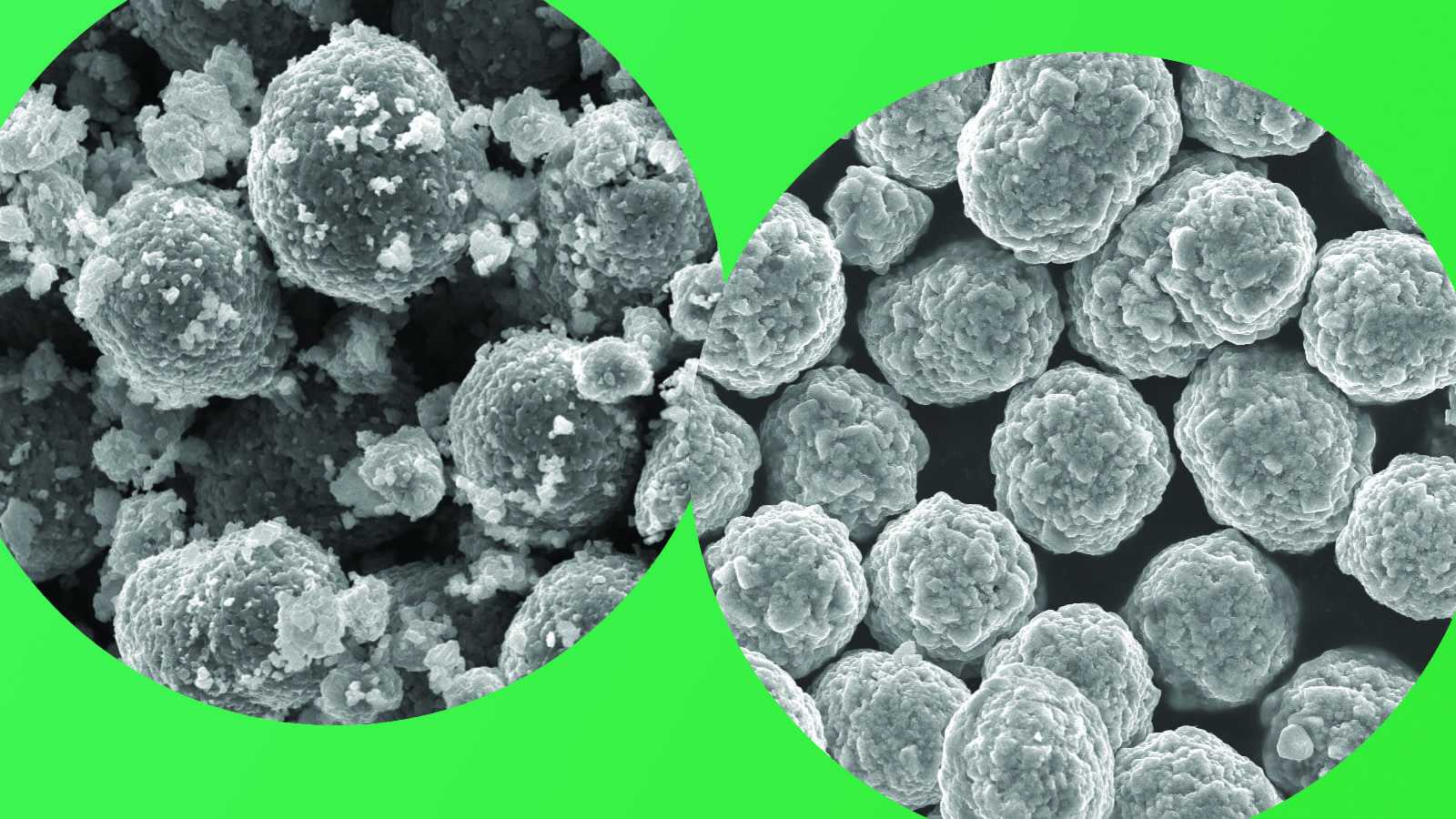
In other research into the materials used in alkaline batteries, the team had obtained data from x-ray scans of batteries made at Brookhaven National Laboratory. They combined the results of their drop tests with the scan data in order evaluate what caused the changes in bouncing.
They found out it had to do with the way the batteries produce power. Electricity is generated by a chemical reaction inside the batteries as zinc changes to zinc oxide. Initially, a layer of zinc surrounds a brass core in the battery like a donut around a hole. As the battery discharges, the zinc donut gradually changes to zinc oxide.
“The zinc oxide begins to form on the outside and it pushes its way to the core,” Steingart said. “As you get more and more zinc oxide, and the zinc oxide begins to appear everywhere in the zinc layer, the battery gets bouncier and bouncier.”
In an article published March 13 in the online version of the “Journal of Materials Chemistry A,” the researchers conclude that the bounces increase because the zinc oxide forms tiny bridges within the zinc material, which decreases the mechanical damping of the battery.
“The zinc starts out as a packed bed of particles that all move very nicely past each other,” Steingart said. “When you oxidize the zinc, it makes bridges between the particles and makes it more like a network of springs. That is what gives the battery its bounce.”
Steingart said that is not too surprising, as zinc oxide is listed as component to add bounce to golf balls in many patents.
But the formation of the bridges reaches a maximum “bounce level” well before the oxidation of the zinc is complete. That means that the bounce will reach a peak and level off well before the battery is actually dead.
“While this test seems a parlor trick, the way in which the coefficient of restitution changes provides startling insight to not only what reaction is occurring into the battery, but where the reaction is occurring as a function of state of charge,” Steingart said.
In addition to Steingart, the researchers on the project included: Shoham Bhadra, a graduate student in the Princeton electrical engineering department and the Andlinger Center for Energy and the Environment was lead author; Benjamin Hertzberg and Andrew Hsieh, post-doctoral researchers in the Princeton mechanical and aerospace engineering department and at the Andlinger Center for Energy and the Environment; Mark Croft of Rutgers University; Joshua Gallaway of City University of New York; Barry Van Tassell of City College of New York; Mylad Chamoun, Can Erdonmez, Zhong Zhong of Brookhaven National Laboratory, and Tal Sholklapper of Voltaiq.
Support for the project was provided in part by the National Science Foundation, the U.S. Department of Energy, and Brookhaven National Laboratory.