Princeton researchers have revealed new links between high-fat diets and aggressive breast cancer, demonstrating the important role fat plays in making some cancers more invasive.
A growing body of evidence has shown that high-fat diets lead to worse outcomes for cancer patients. The Princeton team’s new study, published March 3 in APL Bioengineering, provides a partial answer as to why.
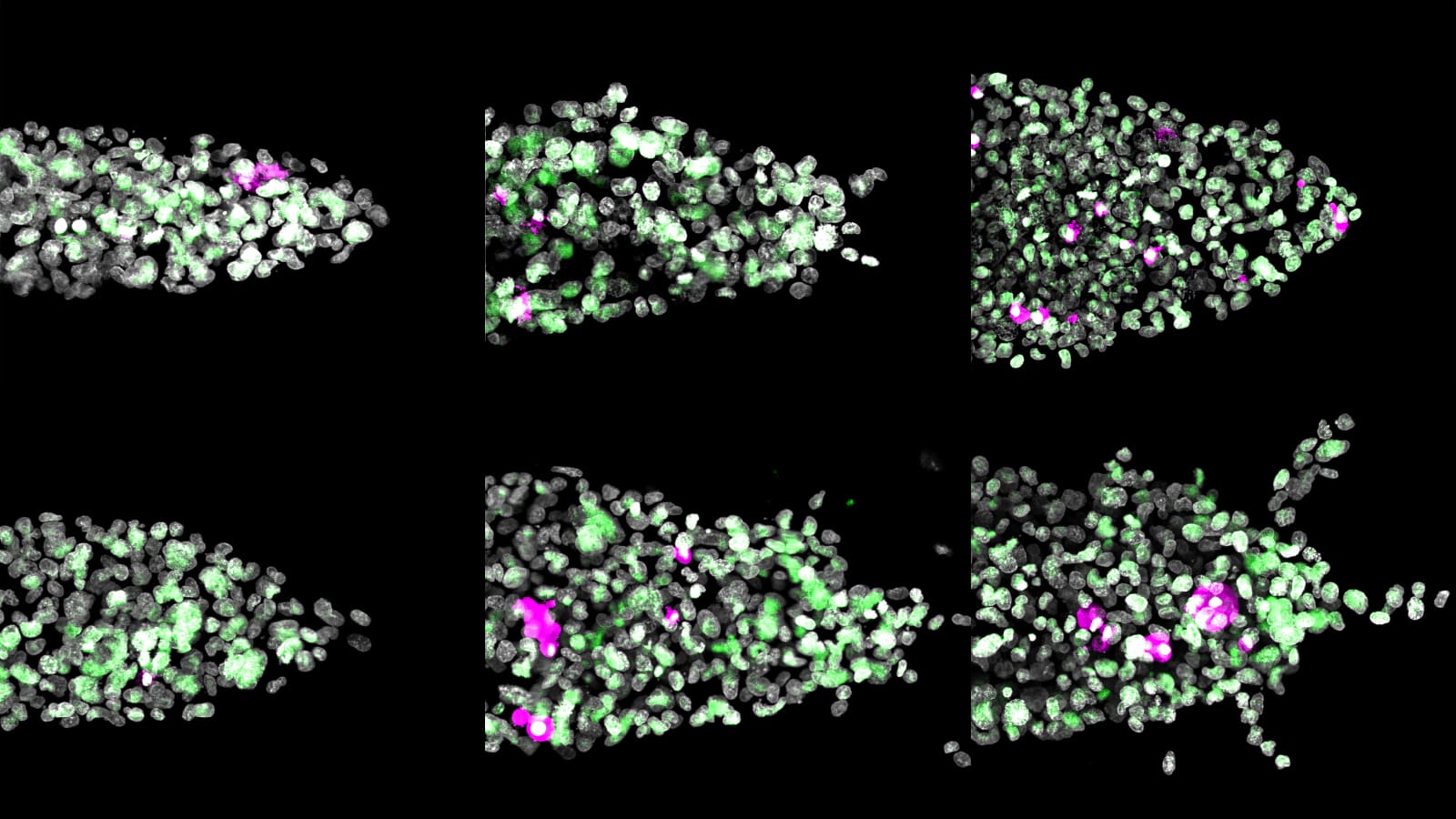
The researchers grew 3D model tumors to test how triple negative breast cancer, a type of cancer that does not respond to most conventional therapies, responds to various diets. They trickled human-like plasma through the tumors to mimic different nutrient conditions in the body. Four diets showed little difference from the baseline — the tumors remained relatively compact. But tumors fed fatty acids and cholesterol formed small, hollow appendages that reached outward, characteristic of aggressive cancers.
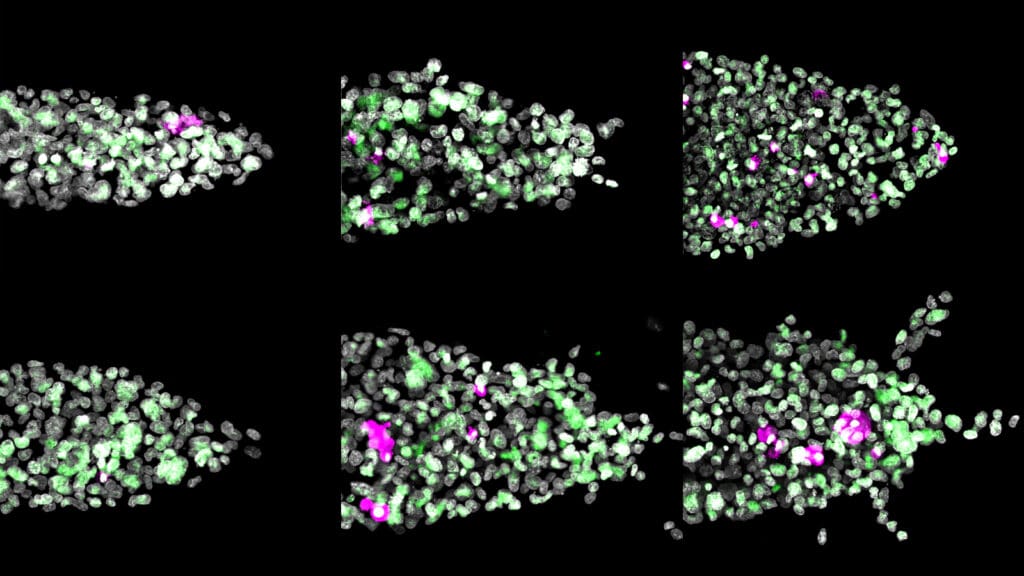
“That’s where the name cancer comes from, crab-like,” said Celeste Nelson, the study’s principal investigator and the Wilke Family Professor in Bioengineering and a professor of chemical and biological engineering. “Aggressive cancers have these tendrils, and it’s the leading edges that end up invading into our normal tissues and making it into either a lymphatic or a blood vessel and escaping and metastasizing.”
Joshua Rabinowitz, professor of chemistry and the Lewis-Sigler Institute for Integrative Genomics and director of the Princeton Branch of the Ludwig Institute for Cancer Research, collaborated on the study.
Tumors studied under conditions including high fat and high insulin
The team grew hundreds of tumors over several years. Tumors fed diets high in insulin, glycerol, and ketones grew about the same as the baseline and did not form hollow invasive cavities.
The high-fat tumors also grew at roughly the same rate as the others, but unlike the others, their structure changed. Cells migrated away from the tumor core and toward its edges. The researchers also saw increased production of a gene associated with breaking down collagen, MMP1, which was strongly correlated with the structural changes.
Nelson said the MMP1 numbers leapt out in the data. The team hypothesized that the high-fat diet played a key role in the gene’s increased expression, promoting degradation of the surrounding environment in a way that presented opportunity for the tumor’s increased aggressiveness, but they have not yet proven that causation. Future studies may look at how high-fat diets influence tumor growth if MMP1 activity is inhibited.
Nelson also cautioned that this study does not necessarily translate to other types of cancer. It does give scientists a new path to study the relationship between diet and prognosis, and a potential target for the development of new therapies.
A ketogenic diet was not helpful
One surprising finding was that the model tumors fed a mix of nutrients designed to simulate a ketogenic diet — a high-fat, low-carbohydrate diet designed to burn calories from fat — did not end up healthier than the baseline, although the findings there were limited.
“We were expecting a ketogenic diet to be protective,” Nelson said. “Yet we didn’t see that here. And it tells us a few possible things. One is that, for this particular type of cancer, maybe a ketogenic diet could be protective, but it operates through other cells that we don’t have in this particular model.”
Although the model more closely resembled human environments than most lab settings, the tumors in this study were still idealized forms that exclude important complex interactions found in the body. Nelson said it could also be that the nature of these tumors is simply too diverse to capture in a single study.
“Every tumor is an individual’s tumor,” Nelson said. “How do you know when you have enough different tumor models to represent the patient population? Maybe that’s not feasible.”
3D cancer model balances complexity and over-simplification
Beyond the main findings, this study also points the way to a more sophisticated study of tumors and their growth than conventional methods provide. 2D cultures in a dish grow in stiff conditions on a diet of simple sugars and electrolytes. Tumors grown in animals, such as mice, introduce so much complexity that it can be hard to isolate the most important interactions. The researchers said that 3D microfluidic models that mimic the tumor’s immediate environment, both physically in terms of the geometry and material properties of surrounding tissues, and chemically in terms of the makeup of bodily fluids and dietary changes, cut a middle path that is more complex and biologically relevant than a 2D dish and more controlled and testable than an animal.
The researchers said this approach is especially good at teasing apart causes of observed effects. For example, ketogenic diets have been shown to delay tumor growth, and this study suggests the effect is mediated through other parts of the tumor environment. That process of elimination can provide rich targets for future studies.
The paper, “Fat promotes growth and invasion in a 3D microfluidic tumor model of triple-negative breast cancer,” was published March 3 in the journal APL Bioengineering. In addition to Nelson and Rabinowitz, the authors include Maryam Kohram, Carolina Trenado-Yuste, Molly C. Brennan-Smith, Evelyn S. Navarro Salazar, Pengfei Zhang, Jasmine E. Hao, Xincheng Xu, Bharvi Chavre, William Oh, Sherry X. Zhang, Susan E. Leggett and Rolf-Peter Ryseck. This work was supported in part by grants from the National Institutes of Health (CA187692, CA214292) and the Ludwig Institute for Cancer Research Princeton.