Stalks, husks and other crop waste show promise as feedstock for sustainable biofuels, but finding microbes that can convert woody plant matter into fuel at industrial scale has proven difficult.
Now, Princeton researchers have taken a step toward engineering a bacterium that converts the material cellulose, found in nearly all plants, into fuels and valuable chemical products. That process requires two steps. The first step is to break the tough cellulose into sugars that a microbe can actually eat. The second step is to eat those sugars and metabolize them into fuel. Few microbes can perform either of these steps efficiently, and currently none can do both at industrial scales.
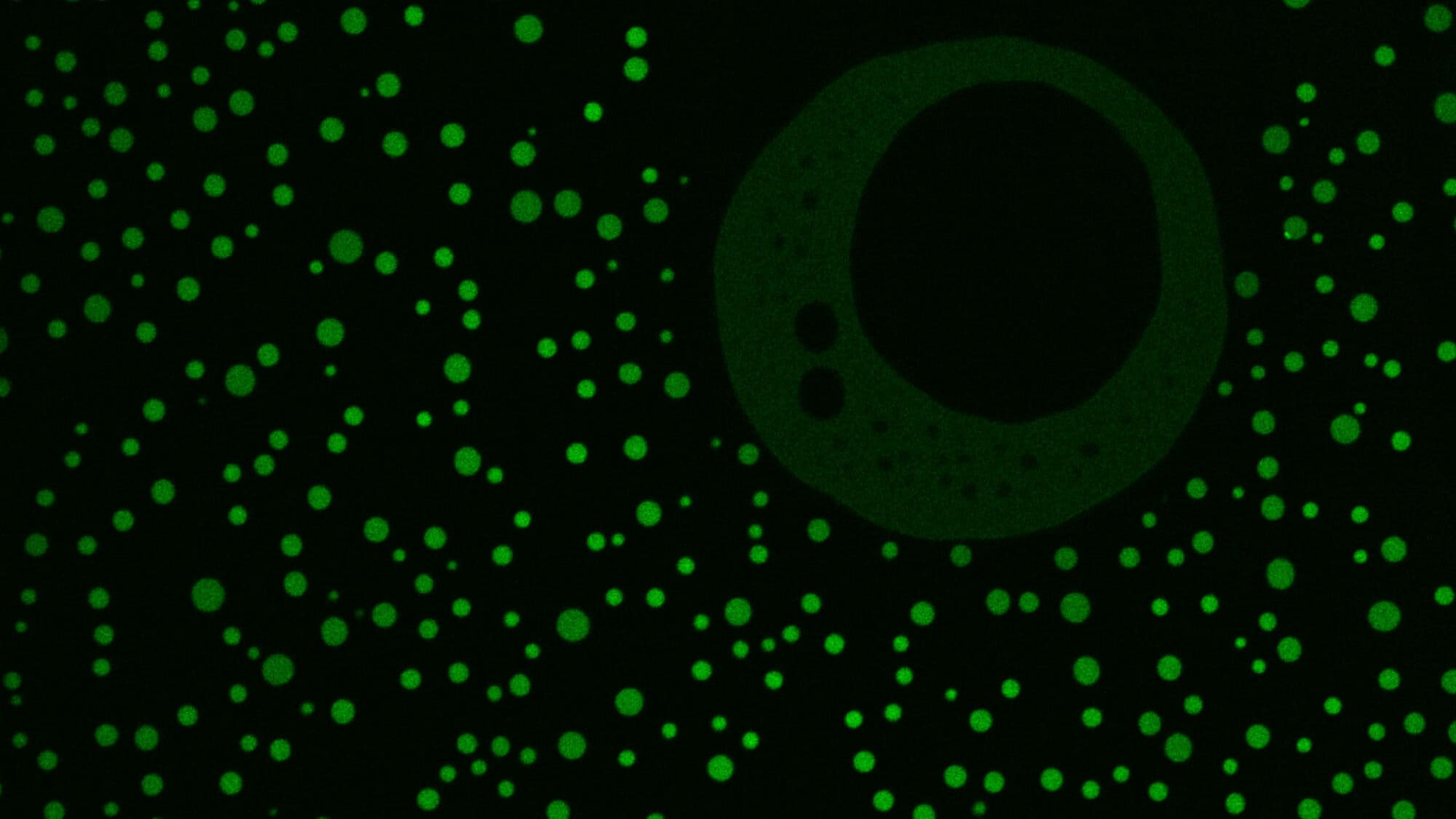
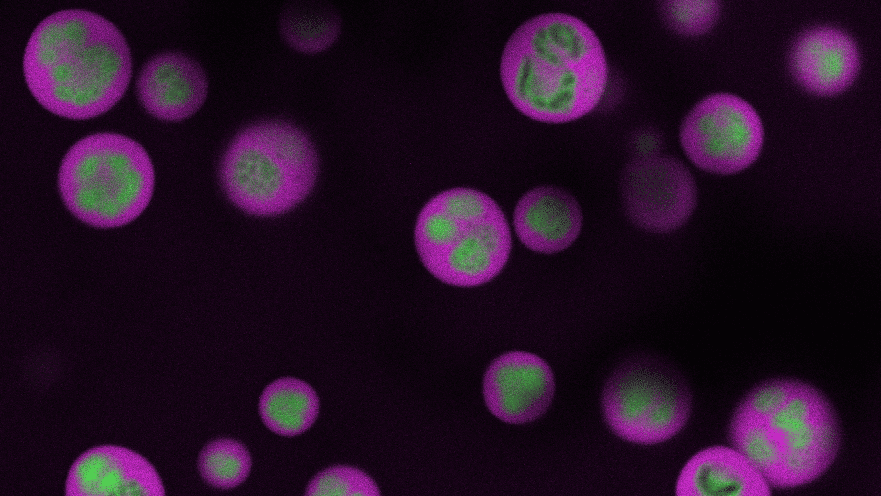
In a study published Jan. 27 in the journal Applied and Environmental Microbiology, the Princeton team looked at the metabolic system of the bacterium Anaerocellum bescii, which can tackle both steps in this process. Previous work showed how this organism releases enzymes that chop the cellulose into bite-sized sugary chunks — a rarefied skill made even more valuable because the microbe thrives in the high temperatures native to bioprocessing systems. The Princeton study identifies the proteins responsible for pulling those chunks inside the microbe’s cell to be metabolized into fuel and shows how editing the organism’s genome can make the microbe even more useful.

“These bacteria are very good cellulose degraders,” said Jonathan Conway, assistant professor of chemical and biological engineering and the study’s principal investigator. “This is the process that they use to eat the sugars from cellulose. It is really central to why they’re useful and interesting.”
To explore this, the researchers deleted the genes they thought were responsible for the transporter proteins that bring cellulose-derived sugars into the cell. The strain missing those genes could eat other sugars but not cellulose, confirming that those proteins are essential for feeding on cellulose.
“The only way for metabolic engineering to really progress is by having a much better sense of what you are trying to engineer in the first place,” said Hansen Tjo, a fifth-year graduate student and the paper’s first author. Understanding the physiology and basic biology of this bacterium, he said, can open the door to engineering the microorganism for industrial applications.

Conway said the larger goal is to find and engineer microbes that can work together in a single system. “Instead of having the enzymes that you make and dump on the plant biomass, and then take the sugar and feed it to yeast, the consolidated bioprocess is to try to put all of that in one pot,” he said. “So, have one organism that makes the enzymes, eats the sugar, makes the product.”

He said in conventional bioprocesses, making the enzymes is often the most expensive part. Anaerocellum bescii is already very good at making enzymes to break the cellulose into edible sugars. This study helps clarify the next step in that process by showing how the bacterium takes up and uses those sugars. It could be possible to engineer two different strains of the bacterium that work together: one focused on producing the enzymes, the other focused on eating the sugars to make the product.
The world generates hundreds of millions of metric tons of crop waste every year, Tjo said. “What if we could turn all that crop waste into sustainable fuels or chemicals instead of relying solely on fossil fuel-based sources?”
The paper, “A highly conserved ABC transporter mediates cello-oligosaccharide uptake in the extremely thermophilic, lignocellulolytic bacterium Anaerocellum bescii(f. Caldicellulosiruptor bescii),” was published Jan. 27, 2026, in the journal Applied and Environmental Microbiology. In addition to Tjo and Conway, the authors include graduate student Virginia Jiang, undergraduate student Anherutowa Calvo, and Jerelle Joseph, assistant professor of chemical and biological engineering.