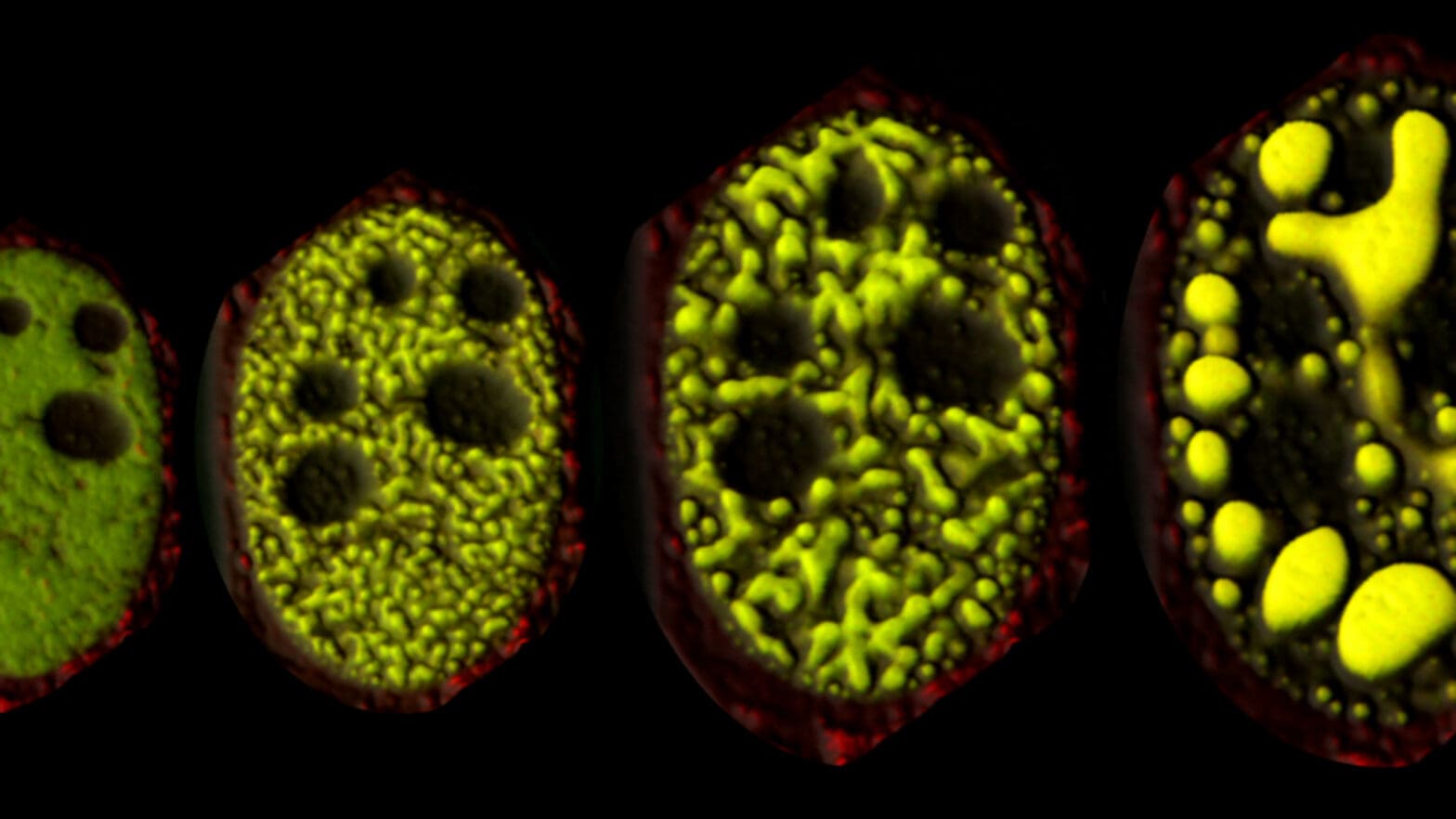
One way that proteins and other molecules in the cell achieve this flexibility is by condensing into specialized droplets, a phenomenon called liquid-liquid phase separation. Clifford Brangwynne, a professor of chemical and biological engineering, leads a research group investigating the formation and functions of these droplets – called membraneless organelles or condensates, in contrast to the better-known membrane-enclosed organelles such as the mitochondria or nucleus.
Membraneless organelles play key roles in the nucleus, where a cell’s genome is stored and its genes are transcribed into messages that direct cellular activities. Brangwynne’s team has developed novel tools to examine and manipulate the condensation of proteins in the nucleus, and is now applying these approaches to uncover new aspects of gene regulation. The work could help elucidate how gene regulation goes awry in cancer and neurodegenerative diseases, and may aid in the discovery of new therapeutic approaches.
“The genome is not just an abstract software program,” said Brangwynne, a Howard Hughes Medical Institute Investigator. Each chromosome is a long strand of DNA that’s packed into a relatively tiny nucleus and wrapped around accompanying proteins. Physical forces are critical to gene regulation, and Brangwynne explores how protein droplets act in, on, and around the genome to influence the expression of genes.
To this end, Brangwynne and colleagues created a technique called CasDrop. It combines optogenetics, or the use of light to control cellular activities, with CRISPR gene-editing technology to activate the condensation of proteins at specific DNA sequences within a cell’s genome. CasDrop allowed the researchers to begin studying the biophysics of the droplets’ interactions with the genome, revealing how protein droplets are attracted to open, active regions of the genome, and how droplets can pull distant parts of the genome together to activate genes in a coordinated fashion.
“These droplets are deforming the genome, and the way in which that’s orchestrated to turn on some genes and turn off others is a very active area of research,” said Brangwynne. “You have many different types of droplets and they can be immiscible with one another – in other words, not able to mix – and you can generate all kinds of interesting structures. We try to think about organization of the genome and expression of the genome and how physical forces associated with these droplets are restructuring it.”