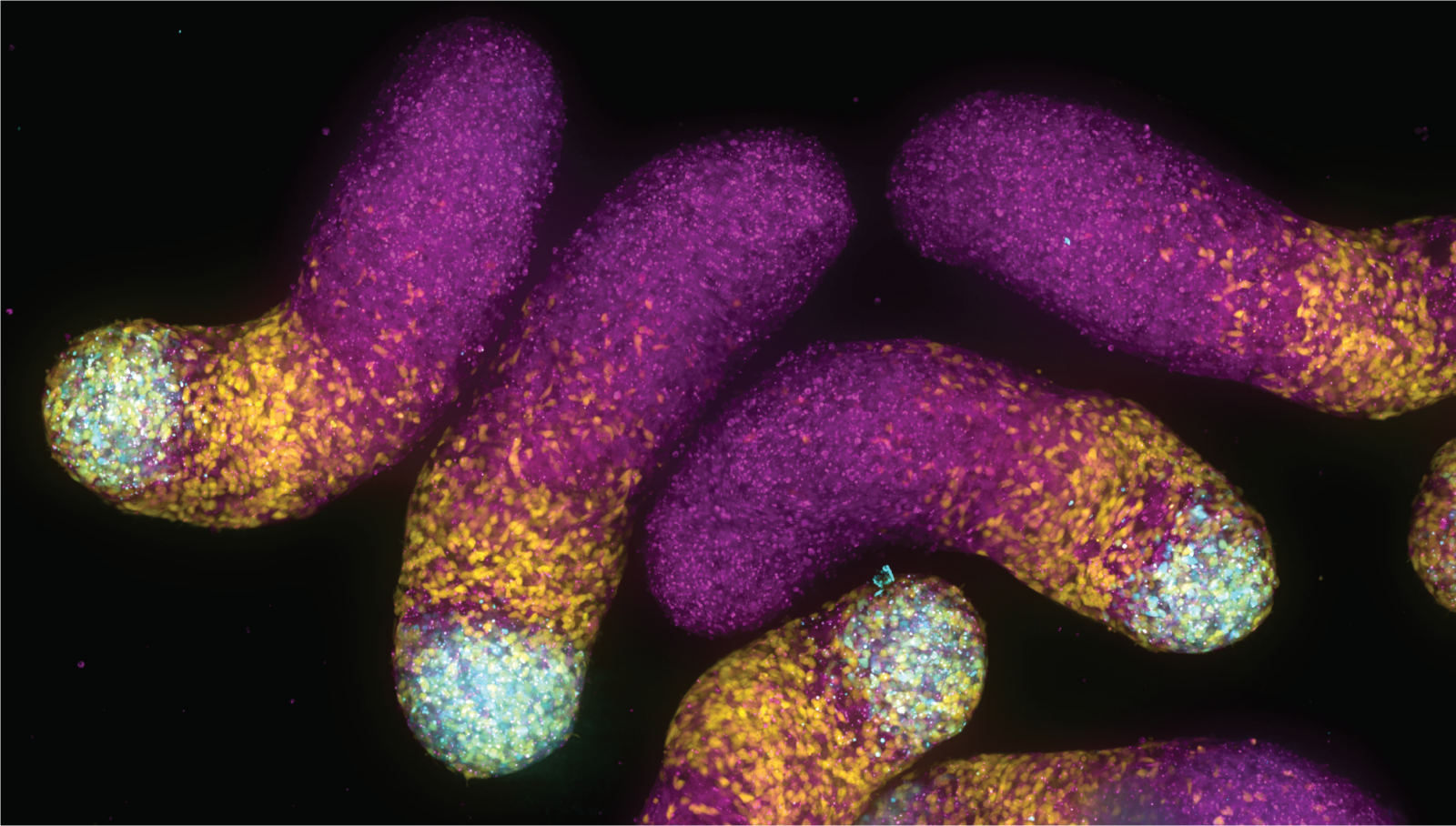
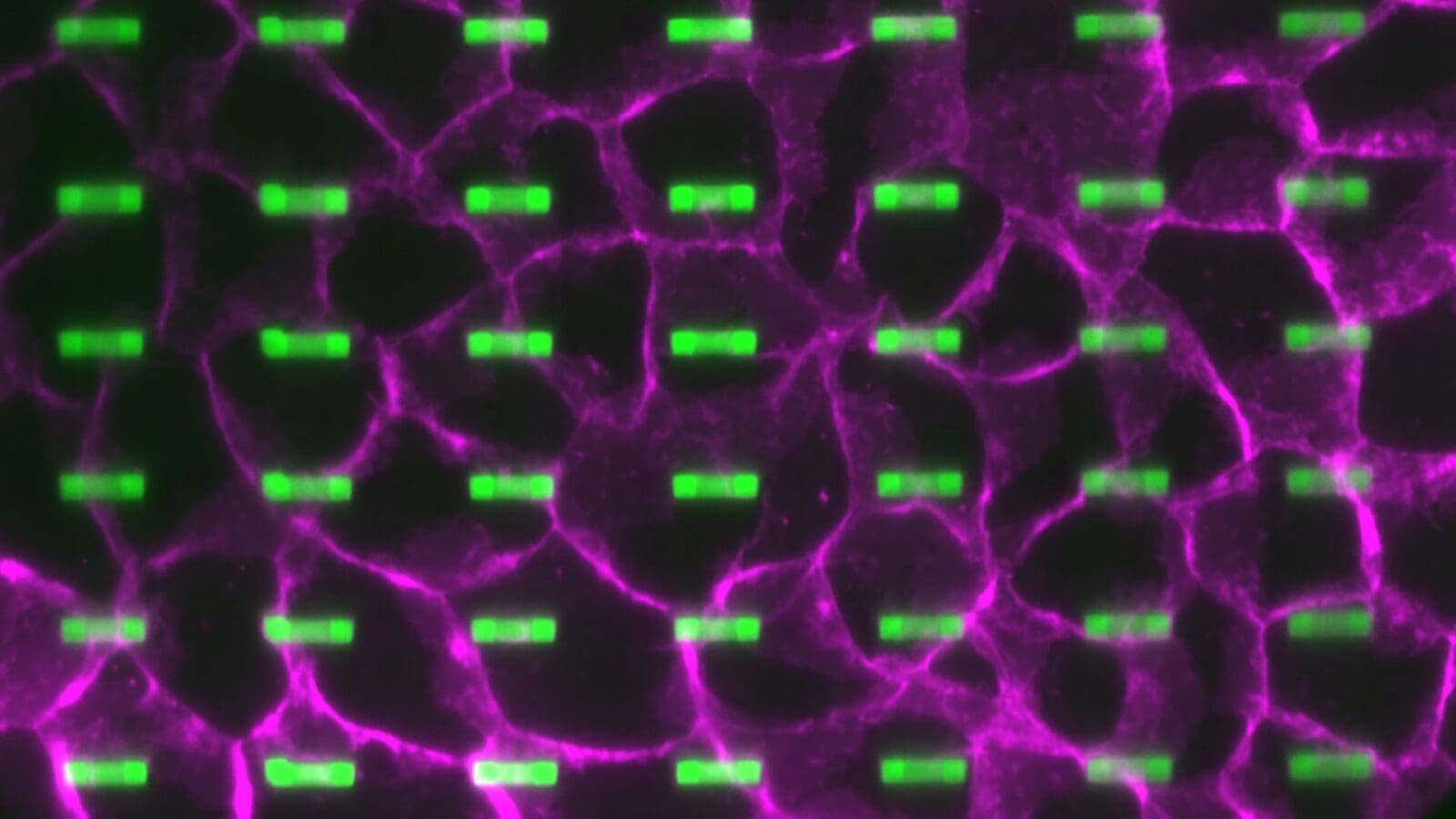
Among other essential functions, these structures direct the life-giving process of cell division and enable immune cells to crawl into wounded tissue.
Now, Princeton researchers who’ve uncovered foundational rules about how these structures form are taking their knowledge a step further. They are building nanorobots inspired by the biology.
“We thought, why only use [our techniques] to understand what the cell is doing? Can we use them to make something that didn’t exist before? Can we create some new technology?” said Sabine Petry, an associate professor of molecular biology.
Petry’s research group has long collaborated with fluid mechanics expert Howard Stone, the Donald R. Dixon ’69 and Elizabeth W. Dixon Professor of Mechanical and Aerospace Engineering, on studies of how tubulin helices form hollow rods called microtubules, as well as their mechanics and their roles in living cells.
Inspired by the extraordinary length and specialized functions of microtubules in nerve cells’ axons — the long cables that carry nerve impulses around the body — Stone and Petry sought to harness microtubules’ circuit-like behavior to build nanomachines, with potential applications in soft robotics, materials science, and medical diagnostics.
In 2021, they received a grant from Princeton’s Eric and Wendy Schmidt
Transformative Technology Fund, and hired two postdoctoral fellows with complementary expertise: Ryungeun Song, a mechanical engineer who had focused on microfluidics in his graduate work; and Meisam Zaferani, a biophysicist who had studied the cues that help mammalian sperm cells navigate toward an egg.
Through simulations and experiments, Song and Zaferani have built devices with narrow channels and tested how different geometries impact the growth of microtubules. From there, they are beginning to test how bespoke architectures can create microtubules that act as useful machines for transporting and sensing molecules on a chip.
“Over a billion years of evolution, biological processes have evolved many different approaches to use these cytoskeletal elements,” said Zaferani, a Gilbert S. Omenn, MD. ’61 and Martha A. Darling *70 Fellow in Molecular Biology. “We’re trying to simulate that using our technology.”
Stone, who frequently collaborates with Princeton colleagues in both engineering and the natural sciences, said, “I find it very interesting to find problems that involve fluid mechanics in other fields, because often
I find a topic that is poorly understood to the scientists on the other side and poorly understood by myself, and together we work
to figure it out.”